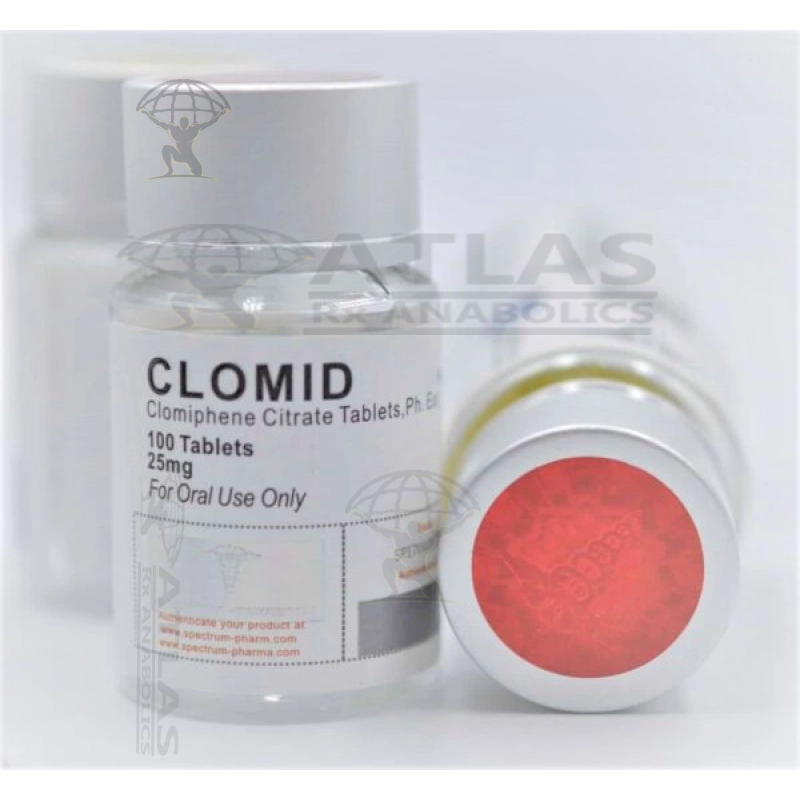
Clomid
- Home
- All products
- Clomid
-
No review yet
Clomid
Quantity
- Categories: LIVEWELL Pharmaceuticals Tablets
FORM 50 mg/tablet 100tablets /bottle Clomid contains a medicine called clomiphene citrate, this belongs to a group of medicines called ovulation stimulants. It works by stimulating the release of eggs ...
Description for Clomid
Clomid, also known by its generic name clomiphene citrate, is a medication commonly used in the field of reproductive medicine. It is primarily prescribed to women who are experiencing fertility issues due to ovulation problems. Clomid works by stimulating the release of hormones necessary for ovulation to occur.
Clomid is available in tablet form and is typically taken orally once a day for a specific duration, as determined by a healthcare professional. The tablets come in different strengths, ranging from 25 mg to 100 mg. The dosage prescribed varies depending on the individual's medical condition and the response to treatment.
When taken, Clomid acts on the pituitary gland in the brain, which then releases follicle-stimulating hormone (FSH) and luteinizing hormone (LH). These hormones are crucial for the development and release of eggs from the ovaries. By increasing the production of FSH and LH, Clomid helps to stimulate ovulation and increase the chances of conception.
Before starting Clomid, it is essential to undergo a thorough medical evaluation to determine the underlying cause of infertility. This may involve assessing hormone levels, conducting imaging tests, and evaluating the overall reproductive health of both partners.
It's important to note that Clomid is not suitable for everyone, and its use should be carefully monitored by a healthcare professional. Some individuals may have certain medical conditions or factors that make them unsuitable candidates for Clomid treatment. Additionally, Clomid should not be used if there is a known or suspected pregnancy, as it may cause harm to the developing fetus.
Like any medication, Clomid may have side effects. Common side effects include hot flashes, mood swings, breast tenderness, nausea, and headaches. These side effects are usually temporary and resolve on their own. However, if any side effects persist or become bothersome, it is important to consult a healthcare provider.
While Clomid is generally well-tolerated, there are some risks associated with its use. These include the potential for ovarian hyperstimulation syndrome (OHSS), multiple pregnancies (such as twins or triplets), and an increased risk of ovarian tumors. Close monitoring by a healthcare professional is essential to minimize these risks and ensure the safe use of Clomid.
In summary, Clomid tablets are a widely used medication for women experiencing ovulation problems and infertility. By stimulating the release of hormones necessary for ovulation, Clomid increases the chances of successful conception. However, it is important to consult with a healthcare professional before starting Clomid and to closely follow their instructions throughout the treatment process.
Based on 0 review(s)
Related Products.
GenLabs CJC-1295 (Without DAC) 5mg Vial with Bacteriostatic Water
In the rigorous world of metabolic and regenerative research, precision, purity, and biological mimicry are paramount. GenLabs introduces its flagship Growth Hormone-Releasing Hormone (GHRH) analog: CJC-1295 (Without DAC) 5mg Vial, presented with a complimentary 2ml vial of USP-grade Bacteriostatic Water. This formulation represents the cutting edge in peptide science, designed for researchers seeking to explore the profound effects of endogenous, pulsatile growth hormone (GH) secretion without the extended half-life modification of Drug Affinity Complex (DAC). For investigations focused on sleep-cycle GH pulses, fat metabolism, cellular repair, and lean tissue accretion, this compound offers an unparalleled tool for accurate physiological modeling.
Understanding the Molecule: CJC-1295 "Mod GRF 1-29"
CJC-1295 is a synthetic analog of the natural Growth Hormone-Releasing Hormone (GHRH, comprising 44 amino acids). The GenLabs variant specifically uses the truncated, more stable, and potent 29-amino acid sequence known in research literature as Mod GRF 1-29. The critical distinction lies in the "Without DAC" designation.
-
With DAC: CJC-1295 with DAC binds irreversibly to albumin in the bloodstream, creating a drug reservoir that releases continuously for days, leading to a sustained, non-physiological elevation of GH and IGF-1.
-
Without DAC (This Product): Our CJC-1295 is without DAC. This means it acts almost identically to the body's native GHRH. It stimulates the pituitary gland’s somatotroph cells, inducing a strong, transient, and pulsatile release of growth hormone that closely mimics the body's natural secretory patterns—particularly the major pulses during deep-wave sleep and post-exercise. This makes it an essential compound for research into circadian hormone rhythms, natural anabolic processes, and targeted metabolic interventions.
Key Research Applications & Mechanistic Pathways
GenLabs CJC-1295 (Without DAC) is utilized in preclinical research models to study:
-
Pulsatile GH Secretion & Circadian Biology: To investigate the role of natural GH pulses in sleep quality, tissue repair overnight, and morning metabolic rates. Its short half-life (~30 minutes) allows for precise timing of pulses, mirroring endogenous cycles.
-
Lipolysis & Body Composition: Research indicates that pulsatile GH release is a potent trigger for lipolysis (fat breakdown). Studies may explore its role in mobilizing stored triglycerides for energy, potentially impacting adipocyte (fat cell) metabolism and reducing visceral fat accumulation.
-
Protein Synthesis & Muscle Recovery: By elevating IGF-1 levels downstream of GH release, this peptide is a tool for examining pathways related to muscle protein synthesis, nitrogen retention, and the repair of musculoskeletal tissues following stress or damage.
-
Cellular Regeneration & Longevity Pathways: GH and IGF-1 play roles in the maintenance of tissues, skin, bone density, and organ health. Research using this precise analog can probe its effects on collagen synthesis, wound healing, and markers of cellular turnover.
-
Synergistic Potentiation: A primary area of advanced research involves the synergistic effect of GHRH analogs with other secretagogues, most notably Ipamorelin. The combination is studied for creating a more potent and selective GH pulse, as Ipamorelin suppresses somatostatin (the hormone that inhibits GH), allowing CJC-1295 to act unopposed.
Why Choose GenLabs CJC-1295 (Without DAC)?
-
Ultra-High Purity (≥99%): Every batch is synthesized using state-of-the-art solid-phase peptide synthesis (SPPS) and purified via HPLC. Each vial includes a Certificate of Analysis (CoA) from an independent, ISO-certified third-party laboratory, confirming identity, purity, and absence of residual solvents or microbial contaminants.
-
Sterile, Lyophilized Powder: The 5mg of active peptide is presented as a sterile, lyophilized (freeze-dried) white powder in a sealed, neutral glass vial. This ensures stability, longevity, and easy reconstitution.
-
Included Bacteriostatic Water (2ml, 0.9% Benzyl Alcohol): We provide a complimentary vial of USP-grade Bacteriostatic Water. The 0.9% benzyl alcohol preserves sterility after reconstitution, allowing multi-dose use when handled correctly under aseptic conditions. This water is specifically formulated for peptide reconstitution.
-
Rigorous Quality Assurance: Manufactured in an ISO 9001 certified facility, our peptides are subjected to Mass Spectrometry (MS) for identity confirmation and HPLC for purity quantification. Vials are filled and capped in a controlled environment.
-
Clarity in Labeling & Use: We provide clear distinctions between "With DAC" and "Without DAC" products to prevent research protocol errors. Our labeling includes batch numbers and expiration dates for full traceability.
Reconstitution, Storage & Handling Protocol
Important Note: This product is for laboratory research use only. It is not for human or veterinary consumption.
-
Reconstitution: Using aseptic technique, gently inject the provided Bacteriostatic Water into the peptide vial. Aim the stream against the glass wall to avoid aggressive agitation. Roll gently between hands until the solution is clear and completely dissolved. Do not shake.
-
Storage: Store the unreconstituted vial in a cool, dark place (optimally at 2-8°C / 36-46°F). After reconstitution, the solution must be refrigerated and used within a defined research timeframe (typically 14-21 days when handled sterilely) due to the absence of preservatives in the peptide vial itself.
-
Dosing Considerations (Research Models): In research settings, dosing is weight-based and timed to mimic natural pulses. Common research protocols involve administration once to twice daily, often in the evening or post-activity. Precise dosing parameters are determined by the specific experimental design.
GenLabs: Your Partner in Advanced Peptide Research
GenLabs is committed to empowering the scientific community with reliable, high-fidelity research compounds. We understand that the integrity of your data begins with the integrity of your reagents. Our CJC-1295 (Without DAC) is more than a peptide; it is a meticulously crafted tool designed to help you uncover the nuanced workings of the somatotropic axis with confidence.
FAQ
Q: What is the difference between CJC-1295 With DAC and Without DAC?
A: The DAC (Drug Affinity Complex) modification creates a long-acting release profile (half-life of several days). "Without DAC" has a short half-life (~30 min), producing a brief, pulsatile GH release that mimics the body's natural pattern.
Q: Why is Bacteriostatic Water included?
A: Bacteriostatic Water contains 0.9% benzyl alcohol, which inhibits bacterial growth. This allows the reconstituted peptide solution to be used multiple times over a short period when stored correctly, reducing waste and cost for research.
Q: What is the purity of this product?
A: Every batch is guaranteed to be ≥99% pure, as verified by independent third-party HPLC analysis. The CoA is available upon request or via our website using the batch number.
Q: How should this be stored long-term?
A: For long-term stability (up to 2 years), keep the sealed, lyophilized vial frozen at or below -20°C (-4°F). For daily use, refrigerated storage is sufficient.
Order Your Research Supply Today
Navigate the complexities of growth hormone research with precision. Select GenLabs CJC-1295 (Without DAC) 5mg Vial for a compound that delivers accuracy, purity, and physiological relevance. Explore our full catalog of synergistic peptides, including Ipamorelin, for comprehensive study designs.
**Live Blend 300mg Steroids: A Comprehensive Overview**
Introduction:
Steroids have gained immense popularity among athletes, bodybuilders, and fitness enthusiasts for their potential to enhance performance and promote muscle growth. One such steroid that has garnered attention is Live Blend 300mg. In this detailed description, we will explore the various aspects of Live Blend 300mg steroids, including its composition, benefits, potential side effects, and usage guidelines.
Composition:
Live Blend 300mg is a combination of three powerful steroids: Testosterone Enanthate, Trenbolone Enanthate, and Drostanolone Enanthate. Each of these compounds plays a unique role in enhancing muscle growth and athletic performance. Testosterone Enanthate is a synthetic variant of the naturally occurring hormone testosterone, which is responsible for muscle development and sexual characteristics. Trenbolone Enanthate is known for its anabolic properties, promoting muscle growth and strength gains. Drostanolone Enanthate, on the other hand, helps in achieving a lean and ripped physique by reducing body fat.
Benefits:
1. Enhanced Muscle Growth: Live Blend 300mg stimulates protein synthesis, leading to increased muscle mass and size. It also promotes nitrogen retention, which is crucial for muscle recovery and growth.
2. Increased Strength and Power: Athletes and bodybuilders who use Live Blend 300mg often experience significant improvements in strength and power. This allows them to lift heavier weights and perform more intense workouts.
3. Improved Endurance: The combination of Testosterone Enanthate, Trenbolone Enanthate, and Drostanolone Enanthate in Live Blend 300mg helps improve endurance levels, allowing users to engage in prolonged and intense physical activities.
4. Fat Loss: Live Blend 300mg has the potential to accelerate fat burning, helping individuals achieve a lean and shredded physique. It increases metabolic rate and promotes the utilization of stored body fat for energy.
5. Enhanced Recovery: Due to its anabolic properties, Live Blend 300mg aids in faster recovery from intense workouts and reduces muscle soreness. This allows athletes to train more frequently and consistently.
Side Effects:
While Live Blend 300mg steroids offer numerous benefits, it is essential to be aware of potential side effects. These may include:
1. Androgenic Effects: Due to the presence of testosterone-based compounds, individuals using Live Blend 300mg may experience androgenic side effects such as acne, oily skin, hair loss, and increased body hair growth.
2. Cardiovascular Risks: Steroids can affect cholesterol levels, leading to an increased risk of cardiovascular issues such as high blood pressure and heart-related diseases.
3. Hormonal Imbalances: The use of Live Blend 300mg can disrupt the body's natural hormone production, potentially leading to testicular atrophy, decreased sperm count, and diminished libido.
4. Liver Toxicity: While Live Blend 300mg is not considered highly hepatotoxic, long-term use or abuse of steroids can put strain on the liver.
Usage Guidelines:
It is crucial to follow proper dosage and usage guidelines while using Live Blend 300mg steroids. These guidelines may vary depending on individual goals, experience level, and tolerance. It is advisable to consult with a healthcare professional or a qualified steroid expert before starting any steroid cycle.
Conclusion:
Live Blend 300mg steroids offer a combination of powerful compounds that can significantly enhance muscle growth, strength, and endurance. While the benefits are appealing, it is essential to understand the potential side effects and follow recommended usage guidelines. Remember, responsible usage and regular monitoring of health are vital to mitigate any risks associated with steroid use.
Bacteriostatic Water 30mL – Sterile, Multi-Dose Diluent for Pharmaceutical and Research Use | GenLabs
Product Code: [Insert Product Code]
Brand: GenLabs
Volume: 30mL Sterile Glass Vial
Preservative: 0.9% Benzyl Alcohol
pH: Balanced to 5.7 (approximately)
Sterility: Sterile Filtered (0.22 micron)
Certifications: cGMP Compliant Manufacturing
Packaging: Sealed, Tamper-Evident Vial with Color-Coded Flip-Off Cap
Introduction: The Gold Standard in Pharmaceutical Reconstitution
In the precise worlds of peptide research, hormone therapy, veterinary medicine, and advanced compounding, the integrity of your active compounds is paramount. However, even the most potent and pure substance is only as reliable as the diluent used to prepare it. Introducing GenLabs Bacteriostatic Water 30mL—the purpose-engineered, sterile solution designed for safe, multi-dose reconstitution and dilution of injectable medications and research peptides.
Bacteriostatic Water is not merely "sterile water." It is a sophisticated pharmaceutical vehicle, meticulously formulated with 0.9% Benzyl Alcohol. This critical additive inhibits microbial growth, allowing a single vial to be used safely for multiple doses over a defined period (typically 28 days), reducing waste and enhancing convenience without compromising sterility. GenLabs manufactures this essential tool under strict cGMP (Current Good Manufacturing Practice) conditions, ensuring every 30mL vial meets uncompromising standards of purity, clarity, and performance. For professionals and informed users who demand precision, this product is the foundational cornerstone of safe and effective solution preparation.
Detailed Product Features & Specifications
1. Pharmaceutical-Grade Sterility and Purity:
The cornerstone of the GenLabs formula is its guaranteed sterility. Each batch is filtered through a 0.22-micron microbiological filter, the industry standard for removing bacteria and potential endotoxins. The water itself is of the highest USP-grade purity, free from pyrogens and particulate contaminants. This ensures that the introduction of the diluent does not introduce any adulterants that could skew research results, cause adverse reactions, or degrade the stability of your reconstituted product.
2. 0.9% Benzyl Alcohol as a Bacteriostatic Agent:
The defining component of bacteriostatic water is the inclusion of Benzyl Alcohol at a concentration of 0.9%. This agent is not an antibacterial in the therapeutic sense but a highly effective bacteriostat. It functions by creating an environment that inhibits the proliferation of most microorganisms that may be inadvertently introduced during a multi-dose withdrawal procedure. This allows for the repeated, aseptic use of the vial for up to 28 days after initial puncture, provided it is stored correctly. It is crucial to note that this formulation is intended for multi-dose use only with compounds that are compatible with Benzyl Alcohol.
3. Optimized pH Balance for Compound Stability:
GenLabs Bacteriostatic Water is carefully adjusted to a slightly acidic pH of approximately 5.7. This is not arbitrary; this pH range is specifically chosen to maximize the stability and solubility of a wide array of peptides and hormonal compounds. A neutral or alkaline pH could lead to accelerated degradation or precipitation of sensitive molecules. Our balanced formula helps maintain the molecular integrity of your reconstituted product from the first dose to the last.
4. Secure, Contamination-Resistant Packaging:
Each 30mL of solution is housed in a sterile, Type I borosilicate glass vial. This pharmaceutical-grade glass is non-reactive, prevents leaching, and ensures the solution remains uncontaminated by the container. The vial is sealed with a sterile bromobutyl rubber stopper, chosen for its superior resealing properties after needle puncture. The entire assembly is secured with an aluminum crimp seal and a distinctive, color-coded flip-off plastic cap, providing a clear, tamper-evident guarantee to the end-user.
5. cGMP Compliance & Traceability:
GenLabs products are manufactured in facilities that adhere to cGMP guidelines. This represents a commitment to quality control far beyond basic standards. It encompasses rigorous documentation, environmental monitoring, raw material testing, and final product release assays. This level of scrutiny provides assurance that every vial is part of a traceable, quality-assured production process.
Primary Applications and Uses
GenLabs Bacteriostatic Water 30mL is an indispensable diluent across multiple fields:
-
Peptide Research: The preferred solvent for reconstituting lyophilized (freeze-dried) research peptides for in vitro or laboratory animal studies. Its bacteriostatic property is essential for multi-dose experimental protocols.
-
Compounding Pharmacy: Used by licensed pharmacists to compound customized injectable medications, including hormone replacement therapies (e.g., testosterone, hCG), pain management solutions, and other sterile preparations.
-
Veterinary Medicine: Employed in veterinary practices for reconstituting vaccines, antibiotics, and hormones for administration to animals.
-
Professional and Educational Settings: Used in laboratory training, medical education, and other settings requiring a reliable, multi-dose sterile diluent for practice or specific procedures.
Important Note: This product is a diluent/solvent only. It contains no active pharmaceutical ingredient (API). It must be used by or under the supervision of qualified individuals who understand sterile technique and the properties of the compound being reconstituted.
Usage Guidelines & Storage Instructions
Proper Reconstitution Technique:
-
Aseptic Hand Hygiene: Begin by thoroughly washing hands and working in a clean, draft-free environment.
-
Sanitize Surfaces: Wipe the rubber stopper of both the Bacteriostatic Water vial and the target compound vial with a fresh 70% isopropyl alcohol swab.
-
Equalize Pressure: Draw a volume of air into your sterile syringe equal to the amount of bacteriostatic water you intend to withdraw. Inject this air into the Bacteriostatic Water vial.
-
Withdraw Diluent: Invert the vial, withdraw the desired volume of water slowly and carefully.
-
Reconstitute: Gently inject the water down the side of the target vial containing the lyophilized powder. Avoid directly spraying onto the powder cake. Swirl gently or roll between hands until the solution is completely clear and the powder is fully dissolved. Do not shake vigorously, as this can cause denaturation of fragile proteins/peptides.
Storage: Store the sealed vial at controlled room temperature (20°-25°C or 68°-77°F), protected from light and freezing. After initial puncture, the vial can be stored for up to 28 days. Always inspect the solution visually before each use; it must be clear, colorless, and free of any particles or cloudiness. Discard if contamination is suspected or after the 28-day window.
Why Choose GenLabs Bacteriostatic Water 30mL?
-
Uncompromised Safety: The 0.9% Benzyl Alcohol formula is the USP-standard for safe multi-dose use, providing a critical defense against microbial contamination.
-
Guaranteed Purity: cGMP manufacturing and 0.22-micron filtration deliver a sterile, pyrogen-free product you can trust.
-
Enhanced Stability: The optimized pH helps preserve the potency and solubility of your valuable reconstituted compounds.
-
Professional-Grade Packaging: Tamper-evident, sterile glass vial with a resealing rubber stopper ensures product integrity from our facility to your point of use.
-
Cost-Effective Convenience: The 30mL multi-dose format minimizes waste, making it an economical and practical choice for ongoing research or treatment protocols.
Frequently Asked Questions (FAQ)
Q: Can this be used for single-dose injections?
A: While it can be, single-dose injections typically require Sterile Water for Injection (SWFI), which contains no preservative. Bacteriostatic Water is specifically designed for multi-dose applications.
Q: Is this safe for human injection?
A: As a sterile, USP-grade pharmaceutical diluent, it is formulated for injection once mixed with a compatible compound. It is NOT for direct intravenous injection as a standalone product. Always follow the guidance of a licensed healthcare provider.
Q: What is the shelf life of an unopened vial?
A: Refer to the expiration date printed on the vial. When stored properly in its sealed condition, it typically has a shelf life of 18-24 months from the date of manufacture.
Q: Why is there a 28-day limit after opening?
A: The bacteriostatic effectiveness of the 0.9% Benzyl Alcohol, combined with the risk of incremental contamination through repeated needle entries, dictates this standard pharmaceutical safety window. Adhering to it is critical.
Q: Is it suitable for all peptides and compounds?
A: Most, but not all. Always verify the compatibility of your specific compound with Benzyl Alcohol. Some specific peptides or individuals with sensitivities may require alternative diluents like Sterile Water for Injection or Sodium Chloride (Bacteriostatic) 0.9%.
Conclusion: The Essential Component for Precision
Reconstitution is the first and most critical step in activating a lyophilized compound. Choosing an inferior diluent risks contamination, instability, and inaccurate dosing. GenLabs Bacteriostatic Water 30mL eliminates this variable, providing a reliable, professionally trusted foundation for your work.
Whether you are advancing a research hypothesis, compounding a tailored medication, or managing veterinary care, ensure your process begins with the gold standard in sterile dilution.
Order GenLabs Bacteriostatic Water 30mL today—where precision meets purity.
Disclaimer: This product is intended for laboratory research, pharmaceutical compounding, and professional use only. It is not for direct human or animal use without being properly compounded with an active ingredient by a qualified professional. The user assumes all responsibility for the safe and appropriate handling and application of this product. Always consult with a healthcare professional or qualified expert for specific guidance.
Tirzepatide 10mg Vial (with Bacteriostatic Water) – Precision Research Compound by GENLABS
In the dynamic landscape of metabolic and endocrine research, the introduction of dual-agonist therapeutics has marked a paradigm shift. GENLABS is proud to supply the scientific community with a critical tool in this arena: Research-Grade Tirzepatide 10mg Vial, provided with a complimentary vial of Bacteriostatic Water (30ml). This offering is meticulously engineered for in-vitro and preclinical studies, providing researchers with a reliable, high-purity compound to investigate the frontiers of receptor pharmacology, weight management science, and glucose homeostasis.
Disclaimer: This product is sold strictly as a chemical or research compound. It is intended for laboratory research purposes only in controlled in-vitro settings. It is NOT for human or veterinary diagnostic or therapeutic use. It is not a drug, dietary supplement, or cosmetic. All information provided is for educational and scientific context. Handle only by qualified, trained research professionals in appropriate laboratory environments.
Unlocking the Dual-Agonist Mechanism: A Scientific Overview
Tirzepatide represents a first-in-class investigational compound engineered as a dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist. This single peptide molecule is designed to mimic the action of both endogenous incretin hormones, which are pivotal in the body's natural regulation of postprandial insulin secretion, appetite signaling, and metabolic efficiency.
-
Dual Receptor Action: Unlike singular GLP-1 agonists, Tirzepatide's novel mechanism engages both the GIP and GLP-1 receptors. Research suggests GIP receptor activation may potentiate the effects of GLP-1, potentially leading to enhanced metabolic outcomes in study models, including more significant impacts on adiposity and hepatic glucose output.
-
Research Applications: This dual action makes our Tirzepatide 10mg vial an invaluable compound for studies focused on:
-
Receptor Binding Kinetics & Signal Transduction: Investigating the affinity, potency, and downstream cellular signaling pathways of dual versus single agonism.
-
Metabolic Pathway Analysis: Studying effects on insulin sensitivity, glucagon secretion suppression, and lipid metabolism in cellular and animal models.
-
Appetite Regulation & Central Nervous System (CNS) Studies: Exploring impacts on hypothalamic appetite centers and satiety signaling pathways in preclinical research.
-
Comparative Efficacy Research: Serving as a benchmark in studies comparing next-generation incretin therapies against earlier compounds.
-
The GENLABS Standard: Uncompromising Purity and Consistency
At GENLABS, we understand that the integrity of your research is directly dependent on the quality of your reagents. Our Tirzepatide is synthesized and controlled under a rigorous quality assurance protocol.
-
High-Purity Synthesis: Produced using advanced solid-phase peptide synthesis (SPPS) techniques, followed by meticulous purification processes.
-
Stringent Analytical Verification: Every batch is verified via High-Performance Liquid Chromatography (HPLC) for purity (typically ≥99%) and Mass Spectrometry (MS) for accurate molecular weight confirmation. These certificates of analysis are maintained for every production lot.
-
Sterile, Lyophilized Form: The peptide is provided as a sterile, lyophilized (freeze-dried) white powder in a sealed, neutral glass vial. This ensures stability, longevity, and ease of precise reconstitution.
-
Complimentary Bacteriostatic Water: Each 10mg vial is paired with a 30ml vial of sterile Bacteriostatic Water (0.9% Benzyl Alcohol). This isotonic, pH-balanced solution contains an antimicrobial agent to allow for multi-dose, stable reconstituted solutions for extended in-vitro use when stored correctly.
Product Specifications & Handling
-
Compound: Tirzepatide (LY3298176)
-
Presentation: 10mg of lyophilized peptide in a sterile glass vial.
-
Molecular Formula: C₂₂₅H₃₄₈N₄₈O₆₈S
-
Molecular Weight: ~4813.5 g/mol
-
Sequence: H-Tyr-*-Glu-Gly-Thr-Phe-Thr-Ser-Asp-Tyr-Ser-Ile-Ala-Met-Asp-Lys-Ile-His-Gln-Tyr-Val-Glu-Phe-Ile-Ala-Trp-Leu-Val-Arg-Gly-Arg-Lys-NH2 (* denotes Aib, α-aminoisobutyric acid)
-
Storage: Store the lyophilized powder at or below -20°C for long-term stability. Upon reconstitution with the provided bacteriostatic water, the solution must be stored refrigerated (2-8°C) and used within a timeframe dictated by sterile lab protocol, not exceeding 30 days due to the preservative.
-
Reconstitution Guide: Aseptic technique is mandatory. Slowly inject the desired volume of bacteriostatic water into the peptide vial. Gently swirl or roll the vial until the powder is fully dissolved. Do not shake violently to prevent peptide denaturation. Final concentration is determined by the researcher based on experimental design.
Intended Research Applications & Pathways
This product is designed for qualified researchers in institutional or accredited laboratory settings. Key research domains include:
-
Biochemistry & Molecular Biology: Studying the structural interaction of Tirzepatide with the GIP and GLP-1 receptors using techniques like surface plasmon resonance (SPR) or crystallography.
-
Cell Biology & In-Vitro Studies: Assessing impacts on insulin secretion in cultured pancreatic beta-cell lines, examining gene expression changes related to metabolism, or probing cellular proliferation/apoptosis in relevant models.
-
Preclinical Metabolic Disease Research: Investigating effects on body weight, adiposity, glucose tolerance, and lipid profiles within established animal models of obesity, metabolic syndrome, or type 2 diabetes, adhering strictly to all ethical and regulatory guidelines (e.g., IACUC protocols).
-
Pharmacology & Toxicology Studies: Establishing dose-response relationships, pharmacokinetic profiles, and safety margins in controlled preclinical models.
Why Choose GENLABS for Your Research Peptides?
-
Commitment to Scientific Integrity: We are a supplier for the research community, not a pharmaceutical retailer. Our documentation, labeling, and communications reflect this scientific focus.
-
Transparency & Documentation: We provide detailed batch-specific analytical data, ensuring you know the exact specifications of the compound in your experiments.
-
Stability & Sourcing: Our cold-chain logistics and direct-from-manufacturer sourcing guarantee the product arrives in optimal condition for your research.
-
Dedicated Support: Our team is equipped to answer technical and specification questions to assist in your procurement process for laboratory supplies.
Important Safety & Legal Notice
This product is for laboratory research use only. It is the sole responsibility of the purchaser to ensure compliance with all local, state, federal, and international laws regarding the purchase, handling, and use of research chemicals. GENLABS products are not designed, manufactured, or intended for human consumption, diagnostic use, or as medicinal products. They must be handled by individuals with appropriate laboratory training and safety equipment, including gloves, goggles, and lab coats.
By purchasing this product, the buyer acknowledges and agrees that they are a qualified researcher or institution operating within a legal and ethical framework for scientific investigation.
YK-11, also known as Myostine, is a synthetic steroidal compound that falls into the category of Selective Androgen Receptor Modulators (SARMs). It was initially developed as a potential treatment for muscle wasting conditions, such as muscular dystrophy. However, it has gained popularity in recent years as a performance-enhancing drug in the bodybuilding and fitness communities.
YK-11 is unique compared to other SARMs because it acts as a partial agonist of the androgen receptor, meaning it binds to and activates the receptor but with a lower affinity than traditional androgens like testosterone. This partial agonism allows YK-11 to selectively stimulate anabolic effects in muscle tissue while minimizing androgenic side effects.
One of the key benefits of YK-11 is its ability to significantly increase muscle mass and strength. It does this by promoting the production of follistatin, a protein that inhibits myostatin. Myostatin is a naturally occurring protein in the body that limits muscle growth. By inhibiting myostatin, YK-11 allows for greater muscle development and improved muscle fiber quality.
Additionally, YK-11 may enhance bone health by increasing bone mineral density and improving bone strength. This can be especially beneficial for individuals dealing with conditions like osteoporosis or those looking to improve their overall bone health.
It is worth noting that, like other SARMs, YK-11 is still under investigation and has not been approved for medical use by regulatory bodies such as the FDA. Consequently, its long-term effects and potential risks are not yet fully understood. Users should exercise caution and consult with a healthcare professional before considering the use of YK-11 or any other performance-enhancing substances.
In summary, YK-11 is a selective androgen receptor modulator that has gained popularity for its potential to enhance muscle growth and strength. While it shows promise in the field of performance enhancement, it is essential to approach its use with caution and seek professional advice to mitigate any potential risks or side effects.
GENLABS TESTOLONE (RAD-140) 10mg | 100 Tablets | The Next Generation of SARM Technology
Product Name: GENLABS TESTOLONE (RAD-140) 10mg Tablets
Quantity: 100 Tablets
Compound: RAD-140 (Testolone)
Dosage: 10mg per tablet
Laboratory: GENLABS
Introducing GENLABS TESTOLONE (RAD-140): Redefining Anabolic Efficiency
In the relentless pursuit of peak physical performance, body recomposition, and metabolic fortitude, the scientific community has long sought an agent that delivers the profound benefits of traditional androgens but without their significant drawbacks. Enter RAD-140 (Testolone), a revolutionary compound at the forefront of Selective Androgen Receptor Modulator (SARM) science. GENLABS is proud to present its pinnacle formulation: TESTOLONE (RAD-140) 10mg Tablets.
This is not merely a supplement; it is a precision-engineered research chemical representing a paradigm shift in how we approach anabolic signaling. Designed for the discerning researcher and disciplined athlete, GENLABS TESTOLONE offers a compelling profile of potent anabolic activity, remarkable tissue selectivity, and a favorable tolerability spectrum. Each bottle contains 100 meticulously dosed 10mg tablets, providing a full cycle for research purposes, crafted with GENLABS' unwavering commitment to purity, accuracy, and pharmaceutical-grade excellence.
What is RAD-140 (Testolone)? The Science of Selectivity
RAD-140, codenamed "Testolone," is a non-steroidal, investigational SARM originally developed for clinical applications requiring anabolic support without estrogenic or androgenic side effects in non-target tissues. Its "selective" mechanism is key to its unique profile.
Unlike traditional anabolic-androgenic steroids (AAS) that bind to androgen receptors (AR) indiscriminately throughout the body—triggering desired effects in muscle and bone but also unwanted effects in the prostate, skin (acne), and scalp (hair loss)—RAD-140 exhibits a high affinity for ARs in skeletal muscle and bone tissue. This targeted approach aims to maximize anabolic activity where it is most desired while minimizing stimulation in tissues associated with side effects. Furthermore, RAD-140 has shown no affinity for converting to estrogen via the aromatase enzyme, eliminating the risk of estrogen-related side effects like gynecomastia and water retention.
Detailed Research Parameters & Expected Observations
Primary Research Applications:
-
Significant Lean Muscle Mass Accretion: RAD-140 is renowned for its potent anabolic capacity. In research models, it promotes a pronounced increase in lean body mass and muscular cross-sectional area. Its mechanism involves powerfully activating AR-mediated pathways in myocytes (muscle cells), leading to enhanced protein synthesis, nitrogen retention, and a profoundly anabolic environment conducive to rapid, quality tissue growth.
-
Exceptional Strength & Power Output: Researchers consistently note dramatic improvements in strength metrics, including maximal force production, power output, and training capacity. This is attributed not only to increased muscle mass but also to RAD-140's potential positive effects on the central nervous system and neuromuscular junction, enhancing neurological drive and muscular contraction efficiency.
-
Enhanced Fat Loss & Metabolic Rate: RAD-140 exhibits a notable ability to promote lipid metabolism and reduce adipose tissue. Its action increases basal metabolic rate (BMR) and promotes a shift in fuel utilization, encouraging the body to prioritize fat as an energy source, thereby supporting sharp, defined physiques and improved body composition ratios.
-
Bone Density & Connective Tissue Support: As a SARM with strong affinity for bone tissue, RAD-140 demonstrates positive effects on bone mineral density (BMD) and skeletal strength. This makes it a compound of interest for research into osteopenia and overall structural integrity. Concurrently, users report enhanced joint resilience and recovery, likely due to increased collagen synthesis and anabolic support in connective tissues.
-
Neuroprotective Properties: Emerging preclinical data indicates that RAD-140 may possess significant neuroprotective effects, potentially offering benefits for cognitive function and neuronal health. This unique attribute separates it from many other compounds in its class.
Why GENLABS TESTOLONE Stands Apart: A Commitment to Uncompromising Quality
In a market rife with inconsistency, GENLABS sets the standard. Our TESTOLONE (RAD-140) 10mg tablets are the product of relentless dedication to scientific integrity.
-
>99% HPLC-Verified Purity: Every batch is independently analyzed using High-Performance Liquid Chromatography (HPLC) to guarantee chemical purity exceeds 99%. No prohormones, no steroids, no hidden blends—just authentic, pharmaceutical-grade RAD-140.
-
Precision Dosing, Guaranteed Potency: Each tablet contains exactly 10mg of active RAD-140. Our state-of-the-art manufacturing ensures perfect dose uniformity, so research results are consistent, predictable, and reliable.
-
Pharmaceutical-Grade Tableting: We utilize advanced direct-compression tableting technology with superior excipients for optimal bioavailability, stability, and dissolution, ensuring the compound is effectively delivered for research.
-
Rigorous Third-Party Laboratory Testing: Beyond in-house QA, samples from every production run are sent to accredited, independent third-party laboratories for verification of identity, purity, and dosage. Certificates of Analysis (CoA) are available, underscoring our commitment to transparency.
-
Secure, Discrete Packaging: Your order arrives in plain, secure packaging, ensuring privacy and product integrity during transit.
Research Guidelines & Important Considerations
-
Research Subject Profile: RAD-140 is typically researched in subjects with a solid training foundation, seeking突破 in body recomposition, strength, and performance.
-
Dosage Protocol: Common research dosages for RAD-140 range from 10mg to 20mg per day, often administered in a single daily dose due to its long half-life (~20 hours). The GENLABS 10mg tablet allows for precise and flexible dosing strategies. A typical research cycle lasts 8-12 weeks.
-
Post-Cycle Therapy (PCT) Consideration: As a potent SARM, RAD-140 can cause significant suppression of endogenous testosterone production via HPTA feedback. While its side effect profile is cleaner than steroids, a comprehensive post-cycle therapy (PCT) protocol, often involving SERMs like Tamoxifen Citrate or Clomiphene Citrate, is considered essential in research to restore natural hormone function after a cycle.
-
Side Effects & Monitoring: Potential research observations may include testosterone suppression, mild lipid profile fluctuations (decrease in HDL cholesterol), and, at higher doses, rare instances of minor androgenic effects. Responsible research includes regular health marker monitoring.
GENLABS: Your Partner in Advanced Performance Research
Choosing GENLABS is choosing a partnership rooted in science, safety, and results. We empower researchers, athletes, and pioneers with the highest-quality compounds, enabling them to explore the boundaries of human performance with confidence.
Disclaimer: GENLABS TESTOLONE (RAD-140) 10mg Tablets are intended for laboratory and research use only, in vitro studies, or athletic performance research not involving human subjects. This product is not a dietary supplement, nor is it intended to diagnose, treat, cure, or prevent any disease. It is strictly for use by qualified research professionals in controlled environments. Keep out of reach of children and unauthorized personnel. By purchasing this product, you acknowledge and certify that you are a licensed researcher and understand the chemical’s intended legal use.
Unlock the next era of anabolic research. Experience the selective power of GENLABS TESTOLONE (RAD-140).
GenLabs Selank (5mg Vial with Bacteriostatic Water): A Professional-Grade Nootropic for Cognitive Equilibrium and Stress Resilience
Meta Title: GenLabs Selank 5mg Vial | Pharmaceutical-Grade Peptide for Anxiety & Focus | With Bacteriostatic Water
Meta Description: Explore GenLabs' premium 5mg Selank vial. Discover how this synthetized nootropic peptide, modeled on Tuftsin, promotes calm focus, enhances memory, and supports stress resilience. Includes bacteriostatic water for precise, sterile reconstitution. For research use only.
Introduction: Redefining Cognitive Calm with Precision Neurochemistry
In the pursuit of optimal mental performance, the modern landscape demands more than just stimulants or simple focus aids. True cognitive excellence arises from a state of balanced equilibrium—where sharp focus coexists with serene calm, and learning is underpinned by emotional resilience. GenLabs introduces its professional-grade Selank (5mg Vial), a synthetized nootropic peptide that represents a paradigm shift in supporting cognitive and emotional well-being. Provided as a pure lyophilized powder with a complimentary vial of Bacteriostatic Water, this kit is engineered for researchers seeking a precise, high-quality tool to explore the intersection of anxiolysis, memory formation, and adaptive stress response.
What is Selank? The Science of a Synthetic Masterpiece
Selank is a laboratory-synthetized heptapeptide (a chain of seven amino acids) with a sequence Thr-Lys-Pro-Arg-Pro-Gly-Pro. Its design is a landmark achievement in peptide science, being a modern analog of the endogenous immunoregulatory peptide, Tuftsin. While sharing a structural lineage, Selank has been strategically modified to enhance its metabolic stability, bioavailability, and, most importantly, its potent effects on the central nervous system.
Unlike many substances that induce either sedation or stimulation, Selank operates through a sophisticated, multi-mechanistic approach to promote homeostasis. Its primary actions are believed to involve modulation of key neurotransmitter systems, including:
-
GABAergic System: Gently influences GABA metabolism, promoting a natural, non-sedating anxiolytic (anti-anxiety) effect without the dulling side effects associated with direct GABA agonists.
-
Serotonergic System: Modulates the expression and utilization of serotonin, a crucial neurotransmitter for mood regulation, contributing to improved emotional balance.
-
Brain-Derived Neurotrophic Factor (BDNF): Research suggests Selank may upregulate BDNF, a protein vital for neuroplasticity—the brain's ability to form new neural connections, learn, and adapt.
This multi-target mechanism results in a unique profile: a clear, calm, and focused mental state conducive to high-level cognitive performance under pressure.
Key Features & Potential Research Applications of GenLabs Selank
GenLabs' Selank is manufactured to exacting standards, ensuring researchers receive a consistent and reliable compound for their investigations.
1. Anxiolytic (Anti-Anxiety) Properties Without Impairment:
Selank’s most documented characteristic is its ability to reduce feelings of anxiety and emotional tension. Crucially, it does this without causing drowsiness, cognitive fog, or dependency. This makes it a compelling subject of study for models of performance anxiety, social stress, and generalized worry, where maintaining mental acuity is paramount.
2. Enhanced Cognitive Function & Memory:
By potentially lowering cortisol (the stress hormone) and supporting BDNF, Selank may create an optimal environment for learning and memory consolidation. Research interest lies in its effects on both working memory (short-term information holding) and long-term memory formation, particularly in subjects under cognitive load.
3. Neuroprotective & Adaptogenic Potential:
Through its influence on stress response systems and neurotrophic factors, Selank may support the brain's resilience against the negative effects of chronic stress. This adaptogenic-like quality positions it as a compound of interest for studying overall neurological health and resilience.
4. Positive Mood Modulation:
By balancing key neurotransmitters, Selank is often associated with a stable, positive mood background. It may support motivation and reduce emotional reactivity, contributing to sustained well-being and productivity.
Product Specifications & GenLabs Quality Guarantee
-
Compound: Selank (Acetylated Heptapeptide)
-
Purity: ≥ 98% (verified by HPLC analysis)
-
Presentation: 5mg of lyophilized (freeze-dried), sterile powder in a sealed glass vial.
-
Included: One 30mL vial of USP-grade Bacteriostatic Water (0.9% Benzyl Alcohol).
-
Storage: Store in a cool, dry place (recommended at 2-8°C / 36-46°F before and after reconstitution).
-
Laboratory Use Only: This product is intended for in-vitro (e.g., cell culture) and research applications in controlled laboratory settings. Not for human or veterinary consumption.
Why the Inclusion of Bacteriostatic Water is Critical: Bacteriostatic Water contains 0.9% Benzyl Alcohol, which inhibits bacterial growth in a multi-dose vial after the rubber stopper is punctured. This allows for safe, sterile, and precise dosing over multiple research sessions, ensuring solution stability and integrity. GenLabs includes this premium diluent to guarantee researchers have all necessary components for proper protocol execution.
The GenLabs Difference: Commitment to Scientific Integrity
Choosing GenLabs means partnering with a brand dedicated to the highest standards of the research community.
-
Third-Party Verification: Every batch is independently analyzed by accredited laboratories, with Certificates of Analysis (CoA) available, confirming identity, purity, and absence of contaminants.
-
Advanced Manufacturing: Produced in ISO-certified facilities using state-of-the-art solid-phase peptide synthesis (SPPS).
-
Stable Lyophilization: Our freeze-drying process ensures long-term stability and potency of the peptide until the moment of reconstitution.
-
Clarity & Transparency: We provide clear product information and emphasize the responsible, legal use of research compounds.
Reconstitution and Handling Guidelines
Disclaimer: The following is a general example for illustrative purposes. Researchers must determine their own specific protocols based on their experimental design.
A common research concentration involves reconstituting the 5mg vial with 1mL of the provided Bacteriostatic Water. This yields a solution with a concentration of 5mg/mL or 5000mcg/mL. Precise, sterile laboratory techniques must always be employed during reconstitution, handling, and storage of the prepared solution to maintain integrity.
FAQ: Addressing Common Research Inquiries
Q: What is the typical research duration for studying Selank's effects?
A: Studies often examine effects over periods ranging from several days to several weeks, noting that benefits may be cumulative.
Q: How does Selank compare to synthetic anxiolytics in research models?
A: Unlike benzodiazepines, Selank's mechanism is indirect and modulating, showing a lower risk profile for tolerance, withdrawal, or significant cognitive impairment in preclinical models.
Q: Can Selank be researched alongside other nootropics?
A: As with any research compound, combining substances should be approached with extreme caution and a deep understanding of potential interactions. Isolated study is always recommended initially.
Q: What is the shelf life?
A: When stored refrigerated and protected from light, the lyophilized powder is stable for up to 24 months. Once reconstituted, the solution should be used promptly and stored under refrigeration for a limited period (typically 7-14 days when using bacteriostatic water).
Conclusion: A Precision Tool for Modern Neuroscience Research
GenLabs' Selank (5mg Vial with Bacteriostatic Water) is more than just a peptide; it is a sophisticated research instrument designed to explore the frontiers of calm cognition. For scientists investigating the neurochemical foundations of anxiety, memory, and stress adaptation, it offers a unique, multi-faceted profile that stands apart from traditional single-target agents.
By delivering pharmaceutical-grade purity, third-party-verified quality, and the essential diluent for proper protocol, GenLabs empowers the research community to conduct rigorous, reproducible science. Discover the potential of balanced neurochemistry with the precision and reliability of GenLabs.
What is LIVEWELL Pharmaceuticals Dianabolan 50?
Dianabolan is an extremely active steroid in terms of the androgenic and anabolic effects on the user. The outcomes of these effects would contribute to a significant rapid increase in muscle mass and strength.
Dianabolan is usually mixed with injectable testosterone drugs, including enanthate, cypionate, and substanon, mostly to start a cycle to increase stamina, muscle weight, and muscle pumping gains before the long-acting testosterone esters listed above become fully effective.
How LIVEWELL Pharmaceuticals Dianabolan 50 works?
Dianabol is a 17-AA steroid. Whether taken on heavy doses or for long periods, it should be remembered that it is liver toxic. The novice athletes should consume 15-40 mg of Dianabol a day, which should not be taken for any longer than 4-6 weeks, and used mostly to initiate the cycle.
For beginners, Dianabolan is a terrible idea, since it is more effective than most steroid injections and the athlete needs a degree at which Dianabol can be used safely in a steroid stack. Dianabolan cycle is a bad idea. It is important to consider the inclination of your body for side effects and it is very important to begin slowly.
How to take LIVEWELL Pharmaceuticals Dianabolan 50
The low-dose use of Dianabolan to initiate a steroid cycle is the most prescribed, and often it is thought to be too much because only testosterone cycles are sufficient to achieve great improvements in both strength and weight of such new steroids.
For the somewhat more mature competitor, Dianabolan 50 is a steroid. Dianabolan only cycles normally require a higher dose of Dianabol to produce favorable outcomes – these cycles are also usually recommended. Less Dianabolan is needed if stacked, as the injection steroid also helps create the necessary muscle properties.
Side effects
Blood work is particularly necessary for Dianabolan before any steroid cycle is started. Where Dianabolan is used for a long time, at an absolute limit of 6 to 8 weeks, blood work should be carried out during the cycle to assess the liver values.
Metandienone side effects include masculinization symptoms, such as acne, increased hair development, voice changes, and increased sexual appetite, estrogenic effects, such as fluid retention, breast enlargement, and liver harm.
How to buy LIVEWELL Pharmaceuticals Dianabolan 50
Dianabolan is a highly active androgenic and anabolic steroid. The results of these effects would help increase muscle mass and strength substantially rapidly. You can buy authentic and safe products from rxanabolics.com.
Shipping Cost
On all orders is set at $25.00
Secure checkout
Protected by Bitcoin
Offer & gift here
On all huge orders